 During this process, the anode experiences corrosion, while the cathode remains unaffected. In practice, linear output currents up to approximately half of this maximum limit can be obtained. The anode and cathode are usually connected via a continuous electrical path while both are immersed in the same electrolyte. 99 were employed as the anode and cathode. Note the only difference is in power pins. Below you can see the 7 segment display pinout for both common anode and cathode 7 segment display. Common Cathode seven segment display’s color is usually black. 1 A simple method of protection connects the metal to be protected to a more easily. Cathodic protection ( CP / kdk / i) is a technique used to control the corrosion of a metal surface by making it the cathode of an electrochemical cell. 
If $\Delta G 0$, the chemical reaction is endergonic, which means it requires external energy to take place.įor electrochemical reactions, every redox pair has its own standard reduction potential $E^$ value will be the reduction, because the reduction reaction for that half reaction is more exergonic. For electrochemical corrosion to occur, three ingredients must be present: an anode, a cathode and an electrolyte. for electrolytic copper refining with soluble anodes to about 5 v. In common cathode seven segment display’s led becomes lit when we apply some +positive voltage on any a,b,c,d,e,f,g pin. Zinc sacrificial anode (rounded object) screwed to the underside of the hull of a small boat. 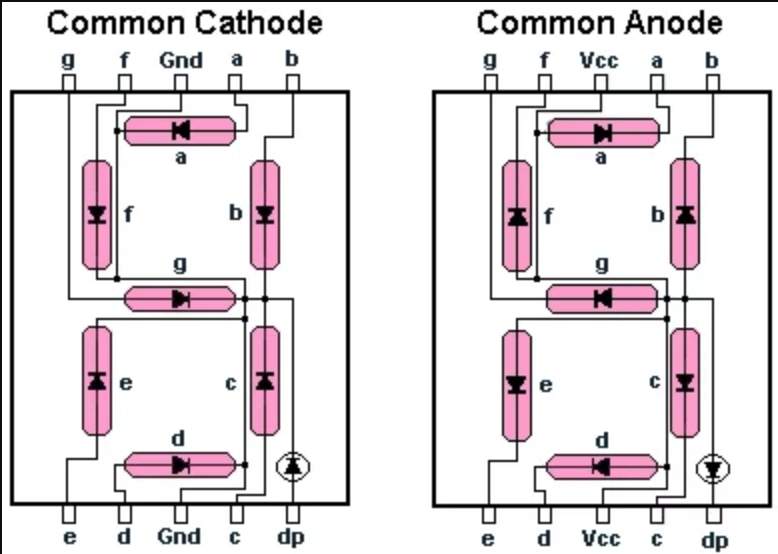
Let's start with thermodynamics: The change in Gibbs free energy $\Delta G$ of a chemical reaction tells you if the reaction can occur without requiring external energy: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
